The Compton effect gives us definitive proof that electromagnetic emission sometimes
can have particle features.
We can ascribe mass to the photon as described by Einstein's famous energy and
mass equivalence equation:
| E = m · c2 = hν | ® | m =
hν/c² m = h/cλ |
c = ν ·λ
A photon has an impulse
p = m · c = (h/cλ)· c
| p = h/l |
Experiments prove the particle features of light. The photon's wave characteristics were also known because of interference and diffraction experiments. Louis de Broglie concluded in 1924 that electrons and protons (and all other particles) can behave like waves. This was proved by electron diffraction and interference experiments in 1927.
According to de Broglie one can write:
| Photon | Particle with mass m = m0/[1 − (v/c)2]½ | |||
| p = | h/λ | = m · v | ® | λ = h/mv |
| E = | hν | = m · c2 | ® | ν = mc²/h |
Focusing from Helium
Light can be diffracted by using Fresnel focusing. Helium atoms can also be diffracted when using an especially thin slit.
| Fig.1: Scanning electron microscope picture of the Fresnel zone plate used to focus helium atoms. The plate has 128 zones and was produced by Heidenhain GmbH, Traunreut, Germany. It is made from a 0.5 µm gold foil; the radius of the innermost zone is 9 µm and the width of the fines ring is 0.4 µm. |
The diffraction experiment with Helium atoms was carried out by group
Toennies (Goettingen). They used the following scheme (Fig.2) in order to shoot a
cooled Helium beam at the grating (Fig.3). The diffracted He-atoms were
detected using a mass spectrometer. The observed He-atoms diffraction (Fig. 4)
directly corresponds de Broglie's predictions. This also made it clear that "wave" model of matter is also valid for compound
systems such as a helium atom. Another group of scientists
have observed the diffraction of Fullerenes by using the double slit experiment.
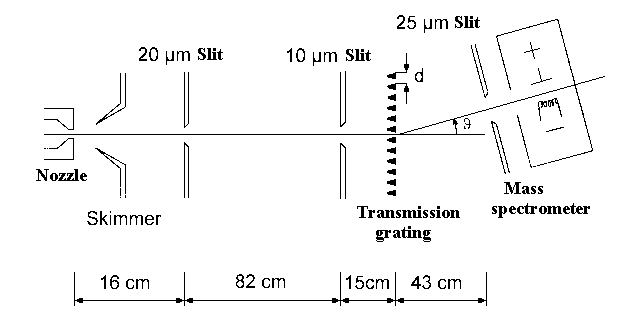 |
| Fig.2: Scientists measured diffraction of He-atoms using this apparatus. |
 |
| Fig.3: Pictured is the diffraction grating which He-atoms cross in order to create an image of their diffraction pattern. One can obtain estimate their sizes from the left picture. The right picture shows the form and production of such a grating. |
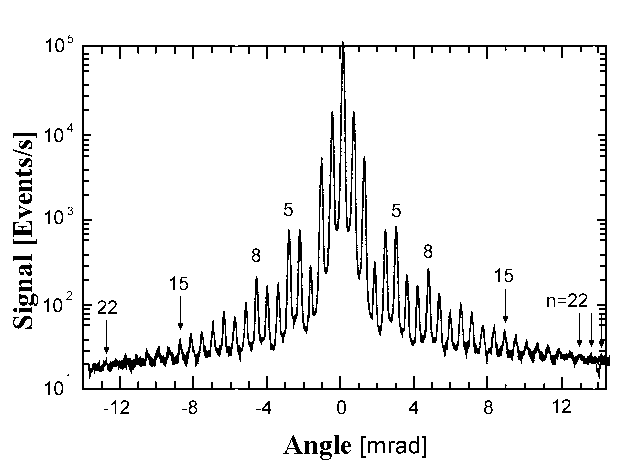 |
| Fig.4: He-atoms produce a signal after passing through mass spectrometer and are detected. The resulting spatial distribution directly corresponds to the de Broglie prediction. This is sufficient proof that compound particles also have wave-like characteristics. |
![]()
Auf diesem Webangebot gilt die Datenschutzerklärung der TU Braunschweig mit Ausnahme der Abschnitte VI, VII und VIII.