Electromagnetic Spectrum
Introduction
For convenience in talking about electromagnetic radiation,
we classify photons of different energies into different spectral regions.
The photons in all of these regions have the same electromagnetic nature,
but because of their very different energies they interact with matter
very differently. For example, the human eye can only detect radiation
that is in the visible region of the spectrum (hence the name). These photons
are both transmitted by the lens of the human eye and absorbed by the photoreceptors
in the retina. There is no fundamental difference in the nature of electromagnetic
radiation of 350 nm versus 400 nm, other than our eyes can sense the 400-nm
photons directly. A 350-nm photon is in the ultraviolet portion of the
electromagnetic spectrum. Some of the boundaries between spectral regions
are not well-defined as between ultraviolet and visible radiation.
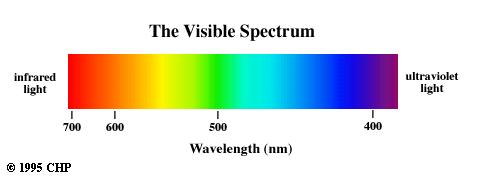
Visible Spectrum
The visible region of the electromagnetic spectrum consists of photons
with wavelengths from approximately 400 to 700 nm. The short wavelength
cutoff is due to absorption by the lens of the eye and the long wavelength
cutoff is due to the decrease in sensitivity of the photoreceptors in the
retina for longer wavelengths. Light at wavelengths longer than 700 nm
can be seen if the light source is intense.
Electromagnetic Spectrum
The following table lists the names of different spectral regions, the
range of frequencies and wavelengths in those regions, and the type of
transition that can occur when a photon in these spectral ranges interacts
with matter.
| Type of Radiation |
Frequency Range (Hz) |
Wavelength Range |
Type of Transition |
| gamma-rays |
1020-1024 |
<1 pm |
nuclear |
| X-rays |
1017-1020 |
1 nm-1 pm |
inner electron |
| ultraviolet |
1015-1017 |
400 nm-1 nm |
outer electron |
| visible |
4-7.5x1014 |
750 nm-400 nm |
outer electron |
| near-infrared |
1x1014-4x1014 |
2.5 µm-750 nm |
outer electron molecular vibrations |
| infrared |
1013-1014 |
25 µm-2.5 µm |
molecular vibrations |
| microwaves |
3x1011-1013 |
1 mm-25 µm |
molecular rotations, electron spin flips* |
| radio waves |
<3x1011 |
>1 mm |
nuclear spin flips* |
*for energy levels split by a magnetic field
Related topics
Auf diesem Webangebot gilt die Datenschutzerklärung der TU Braunschweig mit Ausnahme der Abschnitte VI, VII und VIII.
