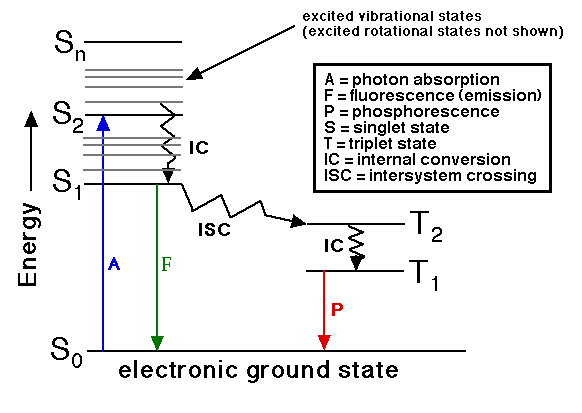
Jablonski diagram:

If the photon emission (shown in short wavelength blue in the diagram) occurs between states of the same spin state (e.g. S1 → S0) this is termed fluorescence. If the spin state of the initial and final energy levels are different (e.g. T1 → S0), the emission is called phosphorescence (shown in longer wavelength red). Since fluorescence is statistically much more likely than phosphorescence, the lifetimes of fluorescent states are very short (1 x 10-5 to 10-8 seconds) and phosphorescence somewhat longer (1 x 10-4 seconds to minutes or even hours; think about glow-in-the-dark flying disks).
Three nonradiative deactivation processes are also significant here:
internal conversion (IC), intersystem crossing (ISC) and vibrational relaxation.
Examples of the first two can be seen in the diagram. Internal conversion
is the radiationless transition between energy states of the same
spin state (compare with fluorescence-a radiative process). Intersystem
crossing is a radiationless transition between different spin states (compare
to phosphorescence). Vibrational relaxation, the most common of the three-for
most molecules, occurs very quickly (<1 x 10-12 seconds)
and is enhanced by physical contact of an excited molecule with other particles
with which energy, in the form of vibrations and rotations, can be transferred
through collisions. This means that most excited state molecules never
emit any energy because in liquid samples the solvent or, in gas phase
samples, other gas phase molecules that are present "steal" the energy
before other deactivation processes can occur.
Auf diesem Webangebot gilt die Datenschutzerklärung der TU Braunschweig mit Ausnahme der Abschnitte VI, VII und VIII.