The intensity of a vibronic transition is proportional to the square of the vibronic transition moment Rv|². In turn, the vibronic transition moment Rv is defined as
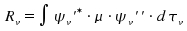
This integration is done over all components of the involved vibrational states. There are two cases.
| Rv | transition |
|---|---|
| equal zero | forbidden |
| not equal zero | allowed |
Simple symmetry considerations suffice to decide whether the integral is zero or not. Provided none of the vibrational states is degenerated, the integrand needs to be totalsymmetric:
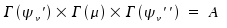
Again, Γ(x) denotes "irreducible representation of x". A indicates the totalsymmetric representation of any point group not degenenerated. If one or both vibrational states are degenerated, the equals sign is replaced by Boole's symbol and we state:
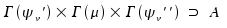
The interpretation of Boole's symbol ⊃ is "contains". For instance, E x E contains A1.
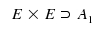
The transition moment is a vector which can be separated into components parallel to the axes x, y and z.
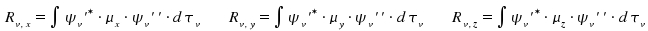
Note that
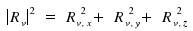
Therefore, a transition v' - v'' is allowed if a least one the the components Rv,x, Rv,y or Rv,z differs from zero.
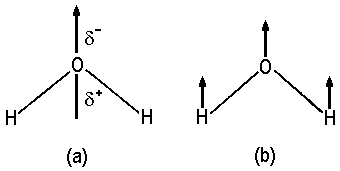 |
| Fig. 1: (a) Vector representing the dipole moment of H2O and (b) Translation of the molecule in this direction. |
Comparable with a translatory motion, the dipole moment is a vector with some direction respective the molecule. In our context, it is necessary to investigate their symmetry properties and, in turn, find the respective irreducible respresentation. For water, this vector belongs to the totalsymmetric representation A1. Generally, the components of the translatory vector and of components of the dipole moment have identical symmetry.
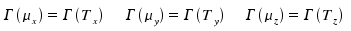
We therefore obtain:
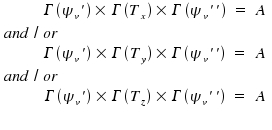
By "and/or", we express that, for an allowed transition, it is sufficient that at least one component of Rv differs from zero.
In most cases, the vibrational ground state with v'' = 0 is the lower state for a transition. The wave function of this state is totalsymmetric, Γ(ψv'') = A, and, as multiplication with a totalsymmetric function leaves everything unaffected, the equations are simplified to
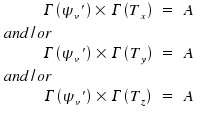
Whenever the totalsymmetric representation is the result of multiplying two representations, there is the conclusion that the two representations had been identical. In consequence
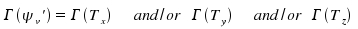
This is the selection rule for a transition from vibrational state v'' = 0 towards other basic oscillations, harmonics, combined oscillations or whatever other not degenerated vibrational state. For a transition from v'' = 0 towards a degenerate state, a analogous selection rule applies.
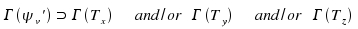
Therefore, the selection rules for any given molecules are determined by the following three steps:
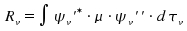
| A1 − A1 | B1 − A1 | B2 − A1 |
These transitions are polarized in direction z, x and y respectively. For instance, an oscillating electric dipol parallel to axis x occurs during a transition B1 − A1.
![]()
Auf diesem Webangebot gilt die Datenschutzerklärung der TU Braunschweig mit Ausnahme der Abschnitte VI, VII und VIII.