R = < yi | a | yj >
where yi and yj are the initial and final states, respectively, and a is the polarizability of the molecule:
a = a0 + (r − re) (da/dr) + .... higher terms
where r is the distance between atoms and ao is the polarizability at the equilibrium bond length, re. Polarizability can be defined as the ease of which an electron cloud can be distorted by an external electric field. Since ao is a constant and
< yi | yj> = 0
R simplifies to:
R = < yi | (r − re) (da/dr) | yj >
The result is that there must be a change in polarizability during the vibration for that vibration to inelastically scatter radiation.
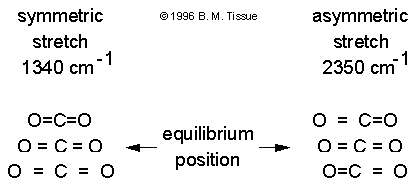
Examples of Raman active and inactive vibrations in CO2

The polarizability depends on how tightly the electrons are bound to the nuclei. In the symmetric stretch the strength of electron binding is different between the minimum and maximum internuclear distances. Therefore the polarizability changes during the vibration and this vibrational mode scatters Raman light (the vibration is Raman active). In the asymmetric stretch the electrons are more easily polarized in the bond that expands but are less easily polarized in the bond that compresses. There is no overall change in polarizability and the asymmetric stretch is Raman inactive.
Raman line intensities are proportional to:
ν4 · σ(ν) · I · exp(−Ei/kT) · C
where ν is the frequency of the incident
radiation, σ(ν) is
the Raman cross section (typically 10-29 cm2), I
is the radiation intensity, exp(-Ei/kT) is the Boltzmann factor
for state i, and C is the analyte concentration.
Auf diesem Webangebot gilt die Datenschutzerklärung der TU Braunschweig mit Ausnahme der Abschnitte VI, VII und VIII.